The FDA Orange Book is the U.S. government’s official list of drugs approved for safety and effectiveness - and it’s the key reason why you can buy a generic version of your prescription for a fraction of the price. Every month, the Food and Drug Administration updates this public database to show which generic drugs are legally allowed to replace brand-name medications. If you’ve ever picked up a pill bottle labeled with a generic name instead of a well-known brand, you’re seeing the Orange Book in action.
What Exactly Is the FDA Orange Book?
The official name is Approved Drug Products with Therapeutic Equivalence Evaluations, but everyone calls it the Orange Book because of its original orange cover. It started in 1984 after Congress passed the Hatch-Waxman Act, a law designed to speed up access to cheaper generic drugs without sacrificing safety. Today, it’s a digital database maintained by the FDA’s Center for Drug Evaluation and Research (CDER). As of late 2023, it listed over 16,000 approved drug products - including both prescription and over-the-counter medicines.
What makes the Orange Book different from a simple list of approved drugs? It doesn’t just say which drugs are approved. It tells you whether a generic version is therapeutically equivalent to the brand-name drug. That means it works the same way in your body, with the same active ingredient, strength, dosage form, and route of administration. This is critical because not all generics are created equal - and the Orange Book is the only place that officially says which ones you can swap without risk.
How Do Generic Drugs Get Listed?
Generic drug makers don’t start from scratch. Instead, they use a shortcut called the Abbreviated New Drug Application (ANDA). To get approval, they must prove their drug is bioequivalent to an already-approved brand-name drug. That brand-name drug is called the Reference Listed Drug (RLD).
For example, if you take Lipitor (atorvastatin), the RLD is the original Pfizer version. Any generic atorvastatin tablet must show it delivers the same amount of medicine into your bloodstream at the same rate as Lipitor. The FDA tests this using blood samples from healthy volunteers. If the results fall within strict limits - usually 80% to 125% of the brand’s performance - the generic is approved.
Once approved, the generic appears in the Orange Book with a clear label: “RLD: No”. The brand-name drug gets marked “RLD: Yes”. This lets pharmacists, doctors, and insurers know exactly which product the generic was tested against. If a generic doesn’t list an RLD, it’s not approved as a substitute.
Therapeutic Equivalence (TE) Codes: The Secret Code Behind Substitution
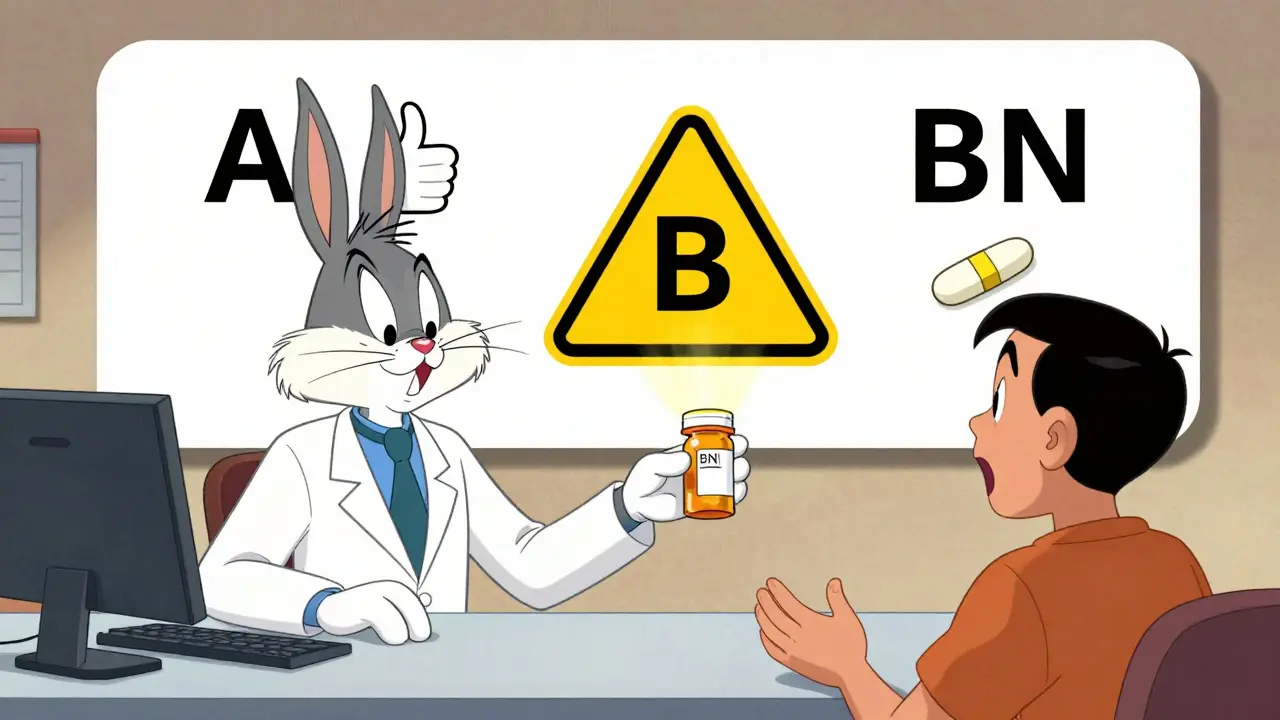
Every approved generic in the Orange Book gets a Therapeutic Equivalence (TE) Code. These two-letter codes tell you whether a generic can be swapped freely with the brand or if there are risks.
- A Code: These generics are considered fully equivalent. You can substitute them without concern. For example, a generic amoxicillin with an “A” code is interchangeable with the brand-name version.
- B Code: These are not recommended for substitution. They might have the same active ingredient but differ in delivery - like a capsule vs. a liquid - or have unproven bioequivalence. Pharmacies can’t switch these without a doctor’s OK.
- BN Code: This means there’s only one manufacturer of the drug on the market. No generics exist yet, so it’s a single-source product.
These codes matter. In 2022, the Congressional Budget Office found that multi-source generics (those with “A” codes) sold for 18-22% less than single-source drugs. That’s the power of competition - and the Orange Book makes it happen.
What About Authorized Generics?
You might see a generic drug that looks exactly like the brand - same color, shape, even packaging - but with a different name. That’s an authorized generic. It’s not made by a competitor. It’s made by the original brand company, sold under a different label at a lower price.
Here’s the catch: authorized generics don’t appear in the Orange Book. They’re listed under the original brand’s New Drug Application (NDA), not as a separate ANDA. The FDA maintains a separate list of authorized generics on its website, updated quarterly. This is important because if you’re comparing prices, you might miss that the “generic” you’re seeing is actually the brand’s own product sold cheaply.
Patents and Exclusivity: The Hidden Rules
The Orange Book isn’t just about generics - it’s also about timing. Brand-name companies must list patents that cover their drug’s active ingredient, formulation, or specific medical use. These patents are published in the Orange Book with a patent number, expiration date, and a “use code” (like U-123) that describes what the patent protects.
When a generic company files an ANDA, it must say whether it’s challenging any of those patents. If it does, the brand company has 45 days to sue. If they do, the FDA can’t approve the generic for 30 months - even if the patent is weak. This is called the “30-month stay.”
Between 2005 and 2022, the number of patents listed in the Orange Book jumped from 8,000 to over 14,000. Critics say this is “patent evergreening” - companies filing minor patents just to delay generics. Supporters say it protects real innovation. Either way, the Orange Book is the battleground.

How Do You Use the Orange Book?
Anyone can access the Electronic Orange Book for free. Here’s how to find what you need:
- Search by brand name - for example, “Lipitor.”
- Look at the active ingredient - “atorvastatin calcium.”
- Find the dosage form and route - like “tablet, oral.”
- Check the RLD column - which product is the original?
- Look at the TE code - is it an “A” or a “B”?
- See if there are multiple generics listed - more competitors usually mean lower prices.
Pharmacists use this daily. A 2022 survey by DRX found that 78% of pharmacy systems integrate Orange Book data directly into their software. But 41% still need extra help interpreting the codes - especially for complex drugs like inhalers or eye drops, where bioequivalence is harder to prove.
What’s Changing in 2025?
The FDA is upgrading the Orange Book. In 2023, it released new rules to stop companies from listing patents that don’t actually protect the drug’s core use. The goal? To cut down on delays caused by “patent thickets” - dozens of weak patents stacked together to block generics.
By 2025, the FDA plans to launch a Digital Orange Book with real-time updates, better search tools, and an API for healthcare systems. Right now, new approvals take about a month to appear. The new system will cut that time in half.
Meanwhile, the FDA is testing more detailed equivalence ratings for complex drugs. For example, two inhalers might have the same active ingredient, but different particle sizes or delivery methods. The current “A” code doesn’t capture that. New codes might soon say “A1” or “A2” to reflect these differences.
Why This Matters to You
If you take a generic drug, the Orange Book is why you can trust it. It’s why your insurance covers it. It’s why you pay $4 instead of $400.
But it’s also why sometimes, a generic doesn’t work the same way for you. Maybe your body reacts differently to the inactive ingredients. Maybe your pharmacist didn’t know the TE code meant “B.” The Orange Book gives you the legal green light - but it doesn’t guarantee perfect results for every person.
For patients, the lesson is simple: if your generic doesn’t feel right, talk to your doctor. For prescribers, it’s knowing the TE code before you write the script. For pharmacies, it’s understanding that the Orange Book is a tool - not a final answer.
The system isn’t perfect. But without the Orange Book, generic drugs wouldn’t exist in the volume they do today. And that’s why it’s one of the most important - and overlooked - tools in American healthcare.
Is the FDA Orange Book the same as Drugs@FDA?
No. The FDA Orange Book only lists drugs that have been approved and are currently on the market with therapeutic equivalence ratings. Drugs@FDA includes all drug applications - including those still under review, tentative approvals, and withdrawn products. You’ll find a generic drug in Drugs@FDA before it appears in the Orange Book, but it won’t be listed as interchangeable until the Orange Book update.
Can a generic drug be pulled from the market after being listed in the Orange Book?
Yes. If a manufacturer fails to meet quality standards, or if new safety data emerges, the FDA can withdraw approval. When that happens, the drug moves to the Discontinued Drug Product List in the Orange Book. It no longer appears in the Active Section and can no longer be legally sold.
Do state laws override the Orange Book’s therapeutic equivalence codes?
Sometimes. Most states allow pharmacists to substitute generics with “A” codes without asking the doctor. But some states have stricter rules - especially for controlled substances or complex delivery systems like inhalers. In those cases, even an “A”-coded drug might require a doctor’s approval. Always check your state’s pharmacy regulations.
Why aren’t over-the-counter (OTC) drugs evaluated for therapeutic equivalence in the Orange Book?
The FDA doesn’t require bioequivalence testing for OTC drugs because they’re generally considered low-risk and widely used without a prescription. The OTC section of the Orange Book simply lists approved products, but it doesn’t assign TE codes. Substitution is left to manufacturers and retailers, not regulators.
How often is the Orange Book updated?
The FDA updates the Electronic Orange Book every month, usually within the first week. New approvals, patent changes, and discontinuations are added then. If a drug is approved on March 10, it will appear in the database by early April. The system doesn’t update in real time - but it’s among the fastest in the industry.





Mantooth Lehto
March 8, 2026 AT 12:59Melba Miller
March 9, 2026 AT 12:01Katy Shamitz
March 10, 2026 AT 03:45Nicholas Gama
March 11, 2026 AT 02:35Mary Beth Brook
March 12, 2026 AT 16:23Neeti Rustagi
March 14, 2026 AT 05:09Dan Mayer
March 14, 2026 AT 23:47Janelle Pearl
March 16, 2026 AT 16:26It’s okay to not feel okay after a switch. You’re not broken. The system just doesn’t account for human variation.
Peter Kovac
March 18, 2026 AT 01:38Erica Santos
March 20, 2026 AT 00:07George Vou
March 20, 2026 AT 05:41Scott Easterling
March 20, 2026 AT 18:39